CBSE Class 11 Chemistry Notes Chapter 5 States of Matter

Class 11 chemistry chapter 5 notes tell us about how things around us look and feel. Everything we touch is called matter. Matter can be a solid like a toy, a liquid like water, or a gas like the air we breathe. These notes help us learn why water flows and why air fills up a balloon by looking at the tiny bits that make them up.
Check Out: Class 11th Books
CBSE Class 11 Chemistry Notes Chapter 5
Matter is the substance that has mass and takes up space. Matter is made up of molecules and atoms. Its chemical and physical characteristics are diverse. There are three categories for matter: solid, liquid, and gas. Their physical qualities arise from variations in the force of interaction that exists within them. Solids interact with the world with the most force, while gases interact with it the least.
Intermolecular Forces
Intermolecular forces are the forces of attraction that exist between molecules, whether they be solid, liquid, or gas. Dipole-dipole, dipole-induced dipole, and dispersion forces add together to form van der Waal forces. Van der Waal forces do not include ion-dipole and ion-induced dipole. The strongest attraction is that of hydrogen bonds.
The Different Types of Intermolecular Forces are
Dipole-Dipole Interaction
Dipole-dipole interactions are seen in polar compounds. Their dipole moments are constant. The molecule attracts both the positive and negative poles. Take HCl, for instance. Cl is more electronegative than hydrogen in the HCl molecule. As a result, the hydrogen atom gains a positive charge whereas the chlorine atom gains a negative charge. Dipole-dipole contact therefore occurs between them.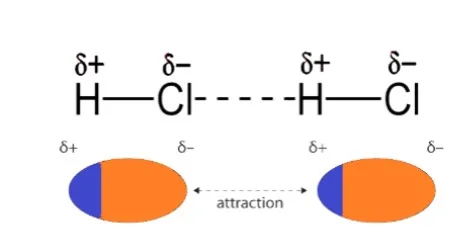
Ion-Dipole Interaction
The attraction between the cation, anion, and a polar molecule is known as ion-dipole interaction. For example, NaCl .
The polar water molecules are attracted towards N a + and C l − on dissolving NaCl in water.
Ion-induced Dipole Interaction
Dipole Induced Dipole Interaction
London forces or Dispersion forces
Check Out: Class 11th Question Banks
State of a Gas and State Variable
The physical condition of the system is the state of a gas. The variables which are used to denote the physical condition of a gas are known as state variables. They are pressure, volume and temperature ( P , V and T ) .Pressure
Pressure is the force applied to an item per unit area. Always apply force in a direction perpendicular to the object. The pascal is the unit of pressure. Numerous instruments are available for measuring pressure. Pressurisation can be measured with a manometer or barometer.Volume
For rigid containers, the volume of the gas is equal to the volume of the container. In non-rigid containers, the number of moles and additional state functions are used to calculate the gas volume.Temperature
Temperature is a physical term that describes how much heat a gas contains. When the temperature of the gas is the same as the surrounding air, no heat is transferred into or out of the gas. The gas's temperature is determined using a thermometer. Fahrenheit, Kelvin, and Celsius are the temperature units.Avogadro’s Law
It provides the relationship between gas volume and quantity. It asserts that there are the same number of molecules in all gases with similar volumes under the same pressure and temperature conditions.
Graham’s Law of Diffusion
Diffusion of gases refers to the intermixing of gases. Even in the absence of pressure differences, gases can combine. If there is a greater pressure differential between the gases, the diffusion process proceeds more quickly. Small apertures allow gas to pass through because of pressure differences. Effusion is the gas flow. Graham's law of diffusion states that there are two variables that affect the rate of diffusion. Gas's molecular weight and partial pressure. The partial pressure and the square root of the gas's molar mass are inversely related to the rate of diffusion, which is directly proportional to both.Real Gases
When considering real gases, the assumption made for ideal gases is no longer applicable. (i) We believed that there are no molecular interactions in an ideal gas. (ii) The volume of a gas's molecules is insignificant in relation to the volume of gases as a whole. In actual gases, the molecule interaction cannot be disregarded. They are as follows: There are long-range attractive forces and short-range repulsive forces. When two real gases are far apart, the interaction forces between them become insignificant. However, as the molecules get closer together, attractive forces begin to form. The molecules begin to reject one another as they get closer.Check Out: PW School Books
Measurement of the Pressure of the Gas
A "barometer" is a common device used to measure a gas's pressure. In a mercury barometer, the atmospheric pressure is measured by the height of a mercury column supported in a sealed glass tube. There are several ways to measure a gas's pressure. Pressure is most commonly measured in terms of height. Assume a liquid of density d 𝑑 is filled in a tube with a cross-sectional area A 𝐴 of height h ℎ . A vacuum has been placed over it. The liquid exerts pressure on the bottom of a container due to gravity.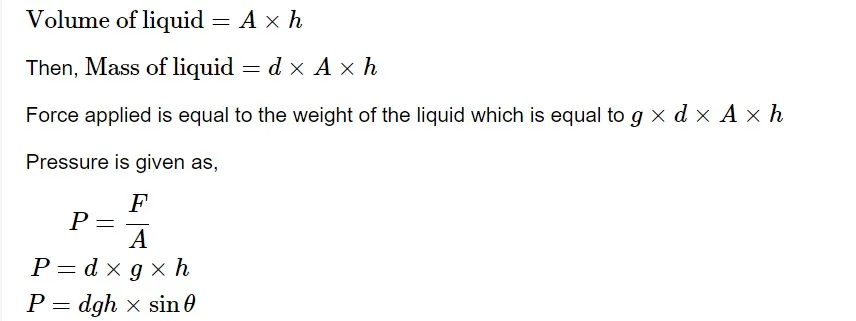


Van Der Waals Equation
After correction of pressure and volume terms, the ideal gas equation can be formulated as,
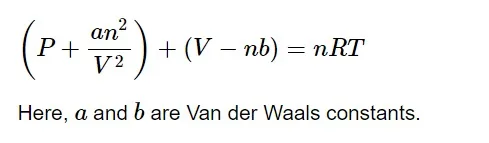
a 𝑎 is the attractive force between the gases. As the attractive forces between the molecules increases, the value of a 𝑎 increases. Whereas, b 𝑏 is the volume occupied by the molecule.
The Van der Waal constant a 𝑎 is always greater than b 𝑏 for a given gas.
The higher the value of a 𝑎 , the more easily liquefaction can occur.
Learning the Basics with Class 11 Chemistry Chapter 5 Notes
Everything is made of very tiny pieces. In class 11 chemistry chapter 5 notes, we learn that how these pieces act decides if something is hard or soft. If the pieces are packed very tight and cannot move, we get a solid. If they can slide around, we get a liquid. If they fly everywhere, we get gas.
The Tiny Pushes and Pulls
-
Intermolecular Forces: These are like tiny magnets that pull the pieces together.
-
Thermal Energy: This is like heat that makes the pieces want to run away from each other.
-
The Big Fight: If the pull is stronger, things stay solid. If the heat is stronger, things turn into gas.
By using class 11 chemistry chapter 5 notes, we can see that gas is the easiest one to study. This is because the tiny pieces are far apart and don't pull on each other much.
Gas Laws and Class 11 Chemistry Chapter 5 Notes PDF
Gases are very funny. If you squeeze them, they get smaller. If you heat them, they get bigger. In the class 11 chemistry chapter 5 notes pdf, scientists have written down "Laws" or rules that gases always follow.
The Simple Rules of Gas
-
Boyle’s Rule: If you squeeze a gas (more pressure), it takes up less space (less volume).
-
Charles’s Rule: If you heat a gas, it needs more space to move around.
-
Gay-Lussac’s Rule: If you heat a gas but keep it in the same box, it pushes harder on the walls.
-
Avogadro’s Rule: More pieces of gas will always take up more space.
If you look at the class 11 chemistry chapter 5 notes pdf, you can find a special math trick called the Ideal Gas Equation. It is written as
PV = nRT
. This one math rule helps us understand how pressure, space, and heat all work together.
How Gases Act in Class 11 Chemistry Chapter 5 Notes
Not all gases are "perfect." Some gases act a bit differently when it gets very cold or very crowded. In class 11 chemistry chapter 5 notes, we learn the difference between "Ideal" gases and "Real" gases.
Why Gases Are Different
-
Ideal Gas: This is a "perfect" gas that follows all rules exactly. It assumes the tiny pieces have no size and don't pull each other at all.
-
Real Gas: Most gases around us are real. When they get squeezed too much, the tiny pieces do take up space and they do start to pull on each other.
The class 11 chemistry chapter 5 notes explain that real gases can turn into liquids if we cool them down and squeeze them. This is how we make things like the gas used for cooking in our kitchens.
Liquids and Class 11 Chemistry Chapter 5 Notes Thermodynamics
Liquids are the middle ground. They have a fixed size but no fixed shape. While some might look for class 11 chemistry chapter 5 notes thermodynamics, those ideas are usually in the next chapter. However, this chapter tells us how liquids act on the surface.
Cool Liquid Facts
-
Vapor Pressure: Even at room temperature, some liquid pieces escape and turn into gas.
-
Surface Tension: The surface of a liquid acts like a stretchy skin. This is why bugs can walk on water!
-
Viscosity: This is a big word for "thickness." Honey is very thick and flows slowly, while water flows fast.
When you study class 11 chemistry chapter 5 notes, you learn that heating a liquid makes it move faster. Eventually, it boils and turns into steam. This happens when the push from the liquid pieces matches the push from the air above it.
Read More: NCERT Solutions for Class 11 Chemistry Chapter 2
Finding Help with Class 11 Chemistry Chapter 5 Notes PDF Download
Studying chemistry can be a lot of fun if you have the right help. You can find a class 11 chemistry chapter 5 notes pdf download to see pictures and easy charts. These charts help you remember the rules for gases and the behavior of liquids very quickly.
Why Use These Notes?
-
Snippet-Friendly: The information is broken into small pieces so you don't get tired.
-
Clear Charts: Tables help you see the difference between solids, liquids, and gases at a glance.
-
Important Points: Only the most important parts are highlighted so you know what to learn first.
|
State of Matter |
Shape |
Volume (Size) |
Movement of Pieces |
|
Solid |
Fixed |
Fixed |
Only vibrate in place |
|
Liquid |
Not Fixed |
Fixed |
Slide past each other |
|
Gas |
Not Fixed |
Not Fixed |
Fly around everywhere |
The class 11 chemistry chapter 5 notes pdf download is great for looking at the "Kinetic Molecular Theory." This is just a way of saying that tiny pieces are always moving.
Books for Class 11
Using good class 11 books makes your study time more useful. Instead of reading the same chapters again and again, you can test what you actually know. Find the books link on the table
|
Book Name |
Link |
|
CBSE Question Bank Class 11 Chemistry |
|
|
CBSE Question Bank Class 11 Combo Set of 4 Books |
|
|
CBSE Class 11 Chapterwise 20 Most Probable |
|
|
CBSE Class 11 Formula Handbook For 2026 Exams |
Class 11 Chemistry Notes Chapter 5 FAQs
Q1: What is the most important rule for gases?
The Ideal Gas Law ( PV = nRT ) is the most important because it combines all the other gas rules into one.
Q2: Why can a bug walk on water?
Because of "Surface Tension," which makes the top of the water act like a thin, stretchy sheet.
Q3: Can we turn any gas into a liquid?
Yes, if we make it very cold and put it under a lot of pressure, the tiny pieces come together to form a liquid.
Q4: What is Viscosity?
It is how much a liquid resists flowing. Honey has high viscosity, while water has low viscosity.
Q5: Is air matter?
Yes! Even though we cannot see it, air takes up space and has weight, so it is matter in a gas state.





