CBSE Class 12 Chemistry Notes Chapter 1 The Solid State

Solid state Chemistry class 12 notes help us learn about why things like stones, wood, and iron are hard. In a solid, the tiny bits inside are packed very close together, like people in a crowded bus. This chapter shows us how these bits are arranged in neat rows and why solids keep their shape.
Check Out: CBSE Class 12 Books
CBSE Class 12 Chemistry Notes Chapter 1 The Solid State
Here we have provided CBSE Class 12 Chemistry Notes Chapter 1 The Solid State-
Solids
Solids possess a definite volume, shape, and mass, which is attributed to the short distance between the particles and the strong interactions that hold them in fixed positions.
Characteristic Properties of the Solid State
(i) Definite Mass, Volume, and Shape: Solids maintain a constant mass, occupy a specific volume, and have a defined shape. (ii) Short Intermolecular Distances: The particles within a solid are closely packed, resulting in minimal space between them. (iii) Strong Intermolecular Forces: The forces of attraction between particles in a solid are strong, keeping the particles tightly bound. (iv) Fixed Particle Positions: The constituent particles (atoms, molecules, or ions) are fixed in position and can only oscillate around their mean positions. (v) Incompressibility and Rigidity: Solids are generally incompressible and maintain their shape without deformation under normal conditions.
Crystal Lattices and Unit Cells
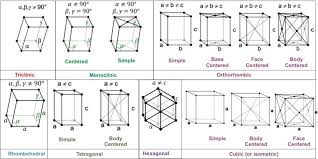
Unit Cell: The unit cell is the smallest repeating unit of a crystal lattice, serving as the fundamental building block of a crystal. By repeating the unit cell in three dimensions, the entire crystal structure is formed.
Types of Unit Cells: A crystal lattice can be generated by repeating a small portion known as the unit cell. There are several varieties of unit cells, each characterized by different arrangements of particles within the cell:
Primitive Cubic Unit Cell:
- Particles are located only at the corners of the cube.
Body-Centered Cubic Unit Cell:
- Particles are located at the corners and one in the center of the cube.
Face-Centered Cubic Unit Cell:
- Particles are located at the corners and at the centers of all the faces of the cube.
Crystal Lattices: A crystal lattice is a three-dimensional arrangement of points representing the positions of atoms, ions, or molecules in a crystal. It forms the framework upon which the crystal structure is built. A crystal lattice is made up of a repeating pattern of unit cells.
Characteristics of Crystal Lattice: (a) Each point in a lattice is referred to as a lattice point or lattice site.
(b) Each lattice point represents one constituent particle, which can be an atom, a molecule, or an ion. (c) Lattice points are connected by straight lines to illustrate the geometry of the lattice, revealing the overall symmetry and structure of the crystal.
Number of Atoms in a Unit Cell
Primitive Cubic Unit Cell
The primitive cubic unit cell has atoms only at its corner. Each atom at a corner is shared between eight adjacent unit cells, four unit cells in the same layer, and four-unit cells in the upper or lower layer. Therefore, only 1/8th of an atom actually belongs to a particular unit cell.
Body-Centred Cubic Unit Cell
A body-centred cubic unit cell has an atom at each of its corners and also one atom at its body centre. Number of Atoms in BCC Cell: Thus, in a BCC cell, we have:
- 8 corners × 1/8 per corner atom = 8 × 1/8 = 1 atom
- 1 body center atom = 1 × 1 = 1 atom
Therefore, the total number of atoms present per unit cell = 2 atoms.
Face-Centred Cubic Unit Cell
A face-centred cubic (FCC) unit cell is structured such that atoms are located at each of the eight corners of the cube and at the center of each of the six faces. The atoms at the face centers are shared between two adjacent unit cells, meaning that only half of each face-centered atom belongs to an individual unit cell.
Number of atoms in BCC cell
a) 8 corners × 18 per corner atom = 8 × 18 = 1 atom b) 6 face-centered atoms × 12 atom per unit cell = 3 atoms Hence, the total number of atoms in a unit cell = 4 atoms Thus, in a face-centred cubic unit cell, we have:
- 8 corners × 1/8 per corner atom = 8 × 1/8 = 1 atom
- 6 face-centered atoms × 1/2 atom per unit cell = 3 atoms
Therefore, the total number of atoms in a unit cell = 4 atoms .
Close-Packed Structures
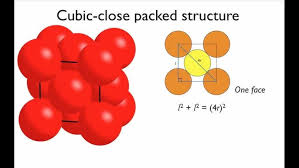 In solids, close packing refers to the arrangement of constituent particles in a manner that minimizes the vacant or empty space within the structure.
In solids, close packing refers to the arrangement of constituent particles in a manner that minimizes the vacant or empty space within the structure.
(a) Close Packing in One Dimension
In one-dimensional close packing, the arrangement of spheres (or atoms) is linear. Here’s how it works:
- Arrangement: Spheres are aligned in a single row where each sphere is directly in contact with its neighboring spheres. This means that each sphere touches two other spheres, one on either side.
- Coordination Number: The coordination number in close packing is defined as the number of nearest neighbor particles that surround a given particle. In one-dimensional close packing, each particle (sphere) has exactly two nearest neighbors — one on the left and one on the right.
(b) Close Packing in Two Dimensions
In two-dimensional close packing, spheres are arranged in a way that maximizes the packing density and minimizes empty space within a plane. Here’s how this can be achieved:
Close Packing in Two Dimensions
In two-dimensional close packing, spheres (or particles) are arranged in a planar pattern where they touch each other. This arrangement can be done in two main ways:
Square Close Packing and Hexagonal Close Packing
Packing in Solids: One and Two Dimensions
Formula of a Compound and Number of Voids Filled
In a close-packed structure, such as in crystalline solids, voids or gaps occur between the constituent particles (atoms, ions, or molecules). These voids are crucial for understanding the packing efficiency and structural properties of the solid. There are two primary types of interstitial voids in a three-dimensional (3D) structure:
Tetrahedral and Octahedral void
Packing Efficiency
Packing Efficiency is the percentage of total space filled by the particles.
Packing Efficiency in hcp and ccp Structures
Hexagonal close packing (hcp) and cubic close packing (ccp) have the same packing efficiency.
Packing Efficiency of a Unit Cell
Efficiency Packing in Body-Centred Cubic Structures
In a body-centred cubic unit cell, one atom is located at the body centre apart from the corners of the cube.
Packing Efficiency in Simple Cubic Lattice
In the simple cubic unit cell, atoms are located at the corners of the cube.
Calculations Involving Unit Cell Dimensions
The unit cell can be seen as a three-dimensional structure containing one or more atoms. We can determine the volume of this unit cell with the knowledge of the dimensions of the unit cell. Mass of unit cell = number of atoms in unit cell × mass of each atom = z × m Where, z = number of atoms in the unit cell, m = Mass of each atom The mass of an atom can be given with the help of Avogadro number and molar mass as: M/N A Where M = molar mass N A = Avogadro’s number The volume of the unit cell, V = a 3 => Density of unit cell = mass of unit cell/ volume of the unit cell => Density of unit cell = m/V = z×ma/a 3 = z×M/a 3 ×N A
Imperfections in Solids
Point Defects:
Point defects in solids are irregularities or deviations from the ideal arrangement of atoms or ions at a single point in the crystal lattice. These defects occur when the crystallization process is too rapid or when there are deviations in the regular pattern of particles. Point defects can significantly impact the properties of the material, such as its density, electrical conductivity, and mechanical strength. They are classified into three main types:
Stoichiometric Defects:
Definition: In stoichiometric defects, the ratio of positive to negative ions in a solid remains unchanged, preserving the overall electrical neutrality of the compound. These defects do not disturb the stoichiometric ratio of the ions but affect the lattice structure.
Types:
- Vacancy Defect: This occurs when an atom or ion is missing from its regular lattice position, creating a vacancy in the crystal structure.
- Interstitial Defect: This defect arises when an extra atom or ion is present in the interstitial spaces (gaps) between the regular lattice points.
Frenkel Defect:
Definition: The Frenkel defect is commonly observed in ionic solids, where a smaller ion (usually a cation) moves from its normal lattice position to an interstitial position. This defect creates both a vacancy (where the ion was originally located) and an interstitial defect (where the ion now resides).
Characteristics: This type of defect maintains electrical neutrality, as the total number of positive and negative charges remains balanced.
Schottky Defect:
- Definition: Schottky defects occur in ionic solids when equal numbers of cations and anions are missing from the lattice, creating vacancies. This type of defect is crucial for maintaining electrical neutrality in the material.
- Characteristics: The loss of ions leads to a decrease in density, and this defect is more common in solids where the sizes of cations and anions are similar.
Electrical Properties
Solids can be classified into three types on the basis of their conductivities. They are: (i) Conductors (ii) Insulators (iii) Semiconductors
Magnetic Properties
Magnetic properties of materials are studied by placing them in a uniform magnetic field and observing their behavior as the magnetic field is varied. The response of a material to a magnetic field depends on its magnetic properties, which are categorized into five major types: (i) Diamagnetic materials (ii) Paramagnetic materials (iii) Ferromagnetic materials (iv) Antiferromagnetic materials (v) Ferrimagnetic materials
Check Out: Class 12th Sample Papers
1. Basics of Solid State Chemistry Class 12 Notes
Everything around you that is firm and hard is solid. In these solid state Chemistry class 12 notes, we see that solids have a fixed shape and a fixed size. The tiny parts inside them cannot move around freely; they can only wiggle or shake in one spot.
-
Strong Pull: The tiny bits pull on each other very hard to stay stuck together.
-
No Squashing: Because the bits are already so close, you cannot squeeze a solid to make it smaller.
-
Slow Movement: Since the bits are stuck, they do not flow like water or fly like air.
-
Weight: Solids usually have a lot of bits in a small space, which makes them heavy.
|
Type of Solid |
How it looks inside |
Examples |
|
Crystalline |
Very neat and tidy rows |
Salt, Sugar, Diamonds |
|
Amorphous |
Messy and random |
Glass, Plastic, Rubber |
2. Using Solid State Chemistry Class 12 Notes PDF
If you use a solid state Chemistry class 12 notes pdf, you will find out that most solids are "Crystalline." This means they are built like a wall made of bricks. Each brick is exactly the same, and they are piled up in a perfect pattern.
The Unit Cell
A unit cell is like a single Lego block. If you keep adding the same block over and over, you get a big tower. In chemistry, the unit cell is the smallest part of the pattern.
Types of Packing
The notes show three ways to pack these tiny bits into a box:
-
Simple Packing: Bits are only at the corners of the box. Most of the box is actually empty air!
-
Middle-Bit Packing (BCC): Bits are at the corners, and one big bit is right in the center of the box.
-
Side-Bit Packing (FCC): Bits are at the corners, and more bits are stuck on the flat faces of the box.
When you look at solid state Chemistry class 12 notes pdf for JEE, you will see that the "Side-Bit" way is the best because it fills up the most space and leaves very little empty room.
3. Empty Spaces and Voids in Hindi
Sometimes it is easier to learn in your own language, so you can look for a solid state Chemistry class 12 notes pdf in Hindi download. This will explain that even if you pack bits very tightly, there will always be tiny holes between them.
-
Voids: These are the empty "holes" or gaps between the bits.
-
Small Holes: A "Tetrahedral" hole is a very tiny gap between four bits.
-
Big Holes: An "Octahedral" hole is a slightly bigger gap between six bits.
-
Fullness: Scientists use math to see how "full" a box is. For example, some boxes are 74% full of bits and 26% full of empty holes.
In the Solid State Chemistry class 12 notes pdf Maharashtra board, you will learn how to calculate the weight of these boxes using the size of the bits and the number of holes.
Check Out: Class 12th Question Banks
4. Mistakes in Solids and Why They Happen
No matter how hard nature tries, it sometimes makes mistakes when building a solid. In our solid state Chemistry class 12 notes, we call these "Defects." These mistakes are actually very useful!
-
Missing Bits: Sometimes a bit is just gone from its spot. This is like a missing brick in a wall.
-
Hiding Bits: Sometimes a bit leaves its seat and goes to hide in one of the tiny "holes" (voids) we talked about.
-
Wrong Bits: Sometimes a different kind of bit (a "stranger") gets into the pattern. This is how we make things like steel or colored gemstones.
-
Color Centers: Some mistakes trap light inside the solid. This is why a clear piece of salt might turn yellow or purple!
5. Electricity and Magnets in Solids for JEE
If you are studying from a solid state Chemistry class 12 notes pdf for JEE, you will learn how solids handle power. This is very important for making things like computers and phones.
-
Conductors: These are solids that let electricity fly through them, like a copper wire or a metal spoon.
-
Insulators: These are solids that block electricity, like a piece of wood or a rubber band.
-
Semiconductors: These are "magic" solids. They only let a little bit of electricity through. We use them to make the brains of our smartphones.
-
Magnetic Solids: Some solids act like magnets. Some pull hard on magnets, and some push them away. It all depends on how the tiny bits inside are spinning.
Whether you follow the solid state Chemistry class 12 notes pdf Maharashtra board or the CBSE rules, remember that the "Solid State" is all about the hidden patterns inside the hard things we use every day.
Also Check: CBSE Class 12 Last 3 Years Previous Year Paper
Class 12 Chemistry Notes Chapter 1 FAQs
1. What is the difference between a crystal and a piece of glass?
A crystal (like salt) has a perfect pattern inside. Glass (which is amorphous) is messy inside and has no real pattern.
2. Why can't I squash a solid like a sponge?
Actually, a sponge has air holes. Real solids have their tiny bits so close together that there is no room to push them any closer.
3. What is a "Void"?
A void is just a fancy word for the tiny empty gaps or "holes" left between the bits in a crystal.
4. Can I study these notes in Hindi?
Yes! You can find a solid state Chemistry class 12 notes pdf in Hindi download to help you understand the topics in a way that feels more natural.
5. Why are semiconductors important in these notes?
They are the most important solids for technology. They help us control electricity, which is how all our electronic gadgets work.
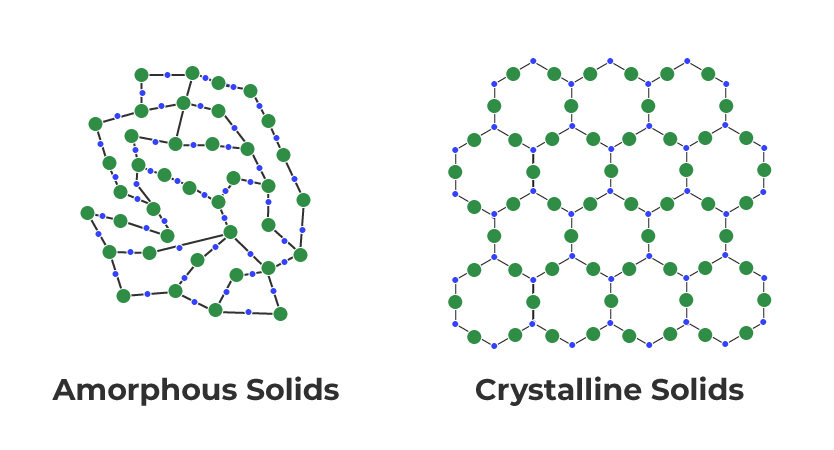 Solids can be classified into two types based on the order of particle arrangement: crystalline and amorphous solids.
Solids can be classified into two types based on the order of particle arrangement: crystalline and amorphous solids.
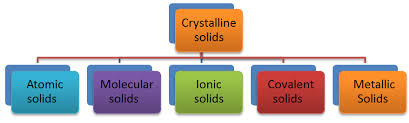 Crystalline solids are classified based on the nature of the interactions between their constituent particles. These interactions determine the properties of the solids, leading to the following four categories:
Crystalline solids are classified based on the nature of the interactions between their constituent particles. These interactions determine the properties of the solids, leading to the following four categories:





