CBSE Class 12 Chemistry Notes Chapter 2 Solutions

Class 12 Chemistry chapter 2 notes explain how we mix two or more things together until they look like one. These mixes are called solutions. In a solution, tiny bits of stuff are spread out so evenly that you cannot see them. This chapter teaches us how to count these bits and see how they change things.
Check Out: CBSE Class 12 Books
CBSE Class 12 Chemistry Notes Chapter 2 Solutions
Here we have provided CBSE Class 12 Chemistry Notes Chapter 2 Solutions-
Solutions
A solution is a homogeneous mixture formed when two or more substances are combined, potentially in different physical states. The substances that make up a solution are known as its components. In a binary solution, which consists of two components, the solvent is the substance present in the larger amount, while the solute is the component present in a smaller amount.
Classification of Solutions:
Based on Physical State:
- Aqueous Solution: If water is the solvent, the solution is called an aqueous solution.
- Non-Aqueous Solution: If the solvent is not water, the solution is termed non-aqueous.
Based on Amount of Solute:
- Unsaturated Solution: This type of solution can dissolve more solute at a given temperature.
- Saturated Solution: In this solution, no more solute can be dissolved at a specific temperature.
- Supersaturated Solution: This contains more solute than what would normally be dissolved at that temperature, often achieved by altering the temperature or pressure.
Solubility
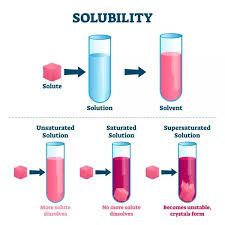
Solubility is defined as the maximum amount of solute that can be dissolved in a specific amount of solvent (usually 100 grams) at a given temperature. It indicates how well a solute dissolves in a solvent to form a solution. The solubility of a solute in a liquid is influenced by several factors:
Nature of the Solute: Different solutes have varying degrees of solubility based on their chemical properties and interactions with the solvent.
Nature of the Solvent: The solvent's properties, such as its polarity and ability to interact with the solute, significantly impact solubility.
Temperature: Solubility generally increases with temperature for most solid solutes, but this can vary for different substances.
Pressure: For gases, solubility in a liquid is directly proportional to the pressure of the gas above the liquid, as described by Henry's Law.
Henry’s Law
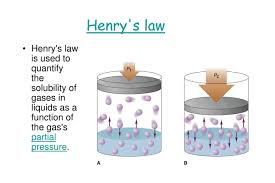
Henry’s Law states that the partial pressure (P) of a gas in the vapor phase is directly proportional to the mole fraction (x) of that gas in the solution.
Applications:
Soft Drinks and Soda Water: Carbon dioxide (CO₂) is added under high pressure to increase its solubility, enhancing the fizz in beverages.
Deep-Sea Diving: To reduce the risk of decompression sickness (the bends), divers breathe a mixture of oxygen and helium. Helium, being less soluble, minimizes the risk of nitrogen bubbles forming in the bloodstream.
High Altitudes: At higher altitudes, the partial pressure of oxygen is lower, resulting in decreased oxygen levels in the blood and leading to conditions like hypoxia or "anoxia," which can affect climbers and travelers at high elevations.
Concentration of Solutions
The concentration of a solution refers to the amount of solute present in a given quantity of solution. It is a measure of how much solute is dissolved in a solvent. Solutions can be categorized based on their concentration into two main types:
Dilute Solution: A dilute solution contains a relatively small amount of solute compared to the solvent. In such solutions, the concentration of the solute is low, meaning that there is a high proportion of solvent relative to solute.
Concentrated Solution: A concentrated solution contains a large amount of solute relative to the solvent. In these solutions, the solute concentration is high, meaning that there is a smaller proportion of solvent compared to solute.
Methods of Expressing Concentration of Solutions
There are several ways to express the concentration of solutions, each suitable for different contexts and precision requirements:
Percentage by Weight (w/w%) : This is the ratio of the weight of the solute to the total weight of the solution, multiplied by 100. It indicates how much of the solute is present in 100 grams of solution.
Percentage by Volume (w/V%) : This can refer either to the weight of the solute in 100 mL of solution or the volume of the solute in 100 mL of solution.
Mole Fraction (x) : This is the ratio of the number of moles of a component to the total number of moles of all components in the solution.
Parts Per Million (ppm) : This measures the amount of solute in one million parts of the solution, useful for trace quantities.
Molarity (M) : This is the number of moles of solute per liter of solution.
Molality (m) : This measures the number of moles of solute per kilogram of solvent.
Normality (N) : This is the number of gram-equivalents of solute per liter of solution.
Formality (F) : This is the number of formula weights of solute per liter of solution.
Mass Fraction : This represents the mass of a component divided by the total mass of the solution, indicating the proportion of the solute in the solution.
Demal (D) : Represents one mole of solute per liter of solution at 0°C.
Raoult’s Law
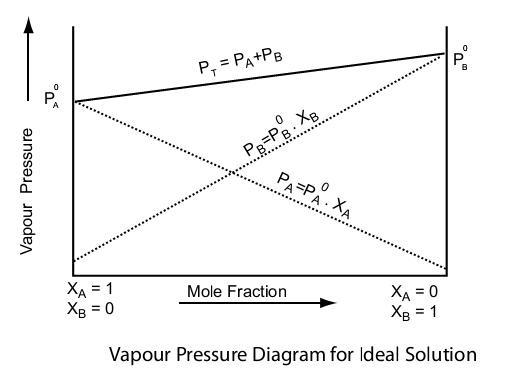 Raoult’s Law describes how the vapor pressure of a solvent is affected when a non-volatile solute is added. The law states that the relative lowering of the vapor pressure of the solvent is directly proportional to the mole fraction of the solute in the solution.
Raoult’s Law describes how the vapor pressure of a solvent is affected when a non-volatile solute is added. The law states that the relative lowering of the vapor pressure of the solvent is directly proportional to the mole fraction of the solute in the solution.
P total = p 1 0 x 1 +p 2 0 x 2
Ideal Solutions
An ideal solution is one that perfectly follows Raoult’s Law across all concentrations. In an ideal solution, the intermolecular forces between the molecules of the solute and solvent are similar to those between the molecules of the pure substances. This means that the interactions between the solute and solvent molecules do not differ significantly from the interactions within each component in its pure state.
Types of Deviations from Raoult’s Law
Positive Deviations:
- Characteristics: In a solution with positive deviations, the vapor pressure of the solution is higher than what Raoult’s Law predicts. This occurs because the intermolecular forces between the solute and solvent molecules are weaker than those between the molecules of the pure substances.
- Reason: The weaker solute-solvent interactions lead to higher vapor pressures because the solute molecules disrupt the intermolecular forces between the solvent molecules, making it easier for molecules to escape into the vapor phase.
- Example: A common example is a mixture of acetone and chloroform. The interaction between acetone and chloroform is weaker than the interactions in pure acetone or pure chloroform.
Negative Deviations:
- Characteristics: In a solution with negative deviations, the vapor pressure of the solution is lower than what Raoult’s Law predicts. This happens because the intermolecular forces between the solute and solvent molecules are stronger than those in the pure substances.
- Reason: Stronger solute-solvent interactions result in a lower vapor pressure. The strong interactions between the solute and solvent molecules reduce the number of molecules escaping into the vapor phase.
- Example: A typical example is a solution of water and hydrochloric acid. The strong hydrogen bonding between water molecules and the ionic interactions with HCl lead to lower vapor pressure than expected.
Konowaloff's Rule
Konowaloff's Rule states that at a fixed temperature, the vapor phase in equilibrium with a solution will always be richer in the more volatile component compared to the solution phase. In simpler terms, the mole fraction of the more volatile component is always greater in the vapor phase than it is in the liquid solution phase.
1. Basics of Class 12 Chemistry Chapter 2 Notes
Everything you drink or breathe is usually a mix. In our class 12 chemistry chapter 2 notes, we learn that every solution has two main parts. One part is the liquid or gas that does the "holding," and the other part is the "stuff" we put inside it.
-
The Solute: This is the small part. It is the stuff that gets mixed in, like salt or a spoonful of sugar.
-
The Solvent: This is the big part. It is the stuff that does the dissolving, like a big glass of water.
-
Mixing Types: You can mix solids into liquids, gases into liquids, or even metals into other metals!
-
Being Even: A true solution is "homogeneous." This means if you take a sip from the top or the bottom, it tastes exactly the same.
|
Part Name |
How much? |
Example |
|
Solute |
Small amount |
Salt |
|
Solvent |
Large amount |
Water |
|
Solution |
The whole mix |
Salt water |
2. Counting Bits in Class 12 Chemistry Chapter 2 Notes Solutions
When you make a drink, it can be weak or very strong. In our class 12 Chemistry chapter 2 notes solutions, we use special math rules to tell exactly how strong a mix is. This is important so that scientists can make medicine or food exactly the right way every time.
Ways to Measure the Mix
-
Weight Percent: This is like a score out of 100. It tells you how many grams of solute are in the whole 100 grams of the mix.
-
Molarity ( M ): This is a very popular way to count. It tells you how many "moles" (which is just a very big number of tiny bits) are in a liter of the drink.
-
Molality ( m ): This is a special way because it uses the weight of the solvent. It is good because it stays the same even if the room gets very hot.
-
Mole Fraction: This is just a simple ratio. It compares the number of "your" bits to the "total" bits in the bowl.
If you get a class 12 Chemistry chapter 2 notes pdf download, you can see all these math rules on one page to help you do your homework.
3. Solubility Rules and Henry's Law
Why does some stuff mix easily while others just sit there? Class 12 Chemistry chapter 2 notes tell us that "like likes like." This means things that are similar want to be friends and mix together.
The Secret of Soda
Have you ever wondered why a soda bottle fizzes when you open it? This is because of Henry's Law.
-
Pressure: If you push down hard on a gas, more of it will hide inside the liquid.
-
Opening the Cap: When you open the soda, the pressure goes away. The gas doesn't want to stay inside anymore, so it jumps out as bubbles!
-
Heat: Most solids (like sugar) dissolve better in hot water. But gases (like the bubbles in soda) actually like cold water much better.
Many students look for Class 12 Chemistry chapter 2 notes solutions to see these rules because they are very common in school tests.
Check Out: Class 12th Question Banks
4. Vapour Pressure and Raoult's Law
When you have a liquid in a closed jar, some of it tries to turn into gas. This makes a tiny "push" on the lid of the jar. We call this vapor pressure. In class 12 Chemistry chapter 2 notes, we see that adding stuff like salt makes this "push" smaller.
The "Perfect" Mix (Raoult's Law)
-
Ideal Solution: This is a perfect mix. The bits like each other just as much as they liked their old friends. They follow the rules exactly.
-
Non-Ideal Solution: These are mixes that do not follow the rules. Sometimes they push harder than they should, and sometimes they push less.
-
Boiling Together: Some mixes are so close that they boil at the same time, as if they were just one thing. These are called Azeotropes.
You might also hear about class 12 Chemistry chapter 2 notes electrochemistry, because the next chapter is about batteries, but for now, we only focus on these liquid mixes.
5. Four Special Rules for Class 12 Chemistry Chapter 2 Notes
This is the most important part of the class 12 Chemistry chapter 2 notes. There are four things that change only when you add more bits, no matter what those bits are. It's like counting how many people are in a room it doesn't matter if they are tall or short, the count is the same!
-
Lowering the Push: When you add salt, the liquid doesn't want to turn into gas as easily.
-
Making it Hotter: Salt water needs more heat to bubble than plain water. This is called "Boiling Point Elevation."
-
Making it Colder: This is how we melt ice on roads. Adding salt makes the water stay liquid even when it is very cold. This is "Freezing Point Depression."
-
The Water Pull: Water likes to move toward the side that has more "stuff" in it. This is called Osmosis. It is how trees pull water up from their roots!
The Van't Hoff Factor ( i )
Sometimes, when you put one bit of salt in water, it breaks into two pieces ( Na and Cl ). We use a special number called " i " to count these extra pieces so our math is correct. You can find this in any class 12 Chemistry chapter 2 notes pdf download.
Also Check: CBSE Class 12 Last 3 Years Previous Year Paper
CBSE Class 12 Chemistry Notes Chapter 2 FAQs
1. What is a "Parts Per Million" (ppm) count?
This is used when there is a very tiny amount of something, like a little bit of dirt in a huge lake. It counts one unit in a million units.
2. Why do we put salt on icy roads?
We do this because of "Freezing Point Depression." Salt makes it harder for water to turn into ice, so the roads stay wet instead of slippery.
3. What is a saturated solution?
It is a "full" mix. If you try to add one more tiny bit of sugar, it will just sink to the bottom because the water can't hold any more.
4. Is there a difference between Molarity and Molality?
Yes! Molarity uses liters (volume), and Molality uses kilograms (weight). Molality is better when the temperature changes.
5. How do I get the class 12 Chemistry Chapter 2 notes pdf download?
You can find it on school help websites and print it out to use as a guide while you study.





