NCERT Solutions for Class 11 Chemistry Chapter 6 Thermodynamics

Finding the right resources for class 11 chemistry chapter 6 NCERT can often feel like trying to compress a gas into an impossibly small container—it requires focus, the right tools, and a clear understanding of the underlying laws. Chapter 6, which focuses on the States of Matter, serves as a bridge between basic chemical concepts and the more complex thermodynamic interactions students encounter later. By utilizing class 11 chemistry chapter 6 ncert solutions, students can navigate the shifts between gases and liquids, understanding why substances behave differently under varying pressures and temperatures. Whether you are looking for the class 11 chemistry chapter 6 ncert to read the core theory or searching for specific class 11 chemistry chapter 6 ncert question answer sets to prepare for exams, having a structured approach is essential. This guide breaks down the chapter’s core complexities, including how these concepts eventually feed into class 11 chemistry chapter 6 ncert solutions thermodynamics, ensuring you have a comprehensive grasp of the material.
Check Out: CBSE Class 11 Books
NCERT Class 11 Chemistry Chapter 6 Exercise Solutions
NCERT Class 11 Chemistry Chapter 6 Exercise Solutions for Thermodynamics help students understand energy changes, enthalpy, entropy, and Gibbs free energy in chemical reactions.
These NCERT solutions make it easier to solve numerical problems and revise key concepts for exams.
Answer the following Questions.
1. Choose the correct answer. A thermodynamic state function is a quantity
(i) used to determine heat changes
(ii) whose value is independent of path
(iii) used to determine pressure volume work
(iv) whose value depends on temperature only
Solution :
A thermodynamic state function is a quantity Whose value is independent of a path. Functions like p, V, T etc. depend only on the state of a system and not on the path.
Hence, alternative (ii) is correct.
2. For the process to occur under adiabatic conditions, the correct condition is:
(i) ∆T = 0
(ii) ∆p = 0
(iii) q = 0
(iv) w = 0
Solution : A system is said to be under adiabatic conditions if there is no exchange Of heat between the system and its surroundings. Hence, under adiabatic conditions, q = 0.
Therefore, alternative (iii) is correct,
3. The enthalpies of all elements in their standard states are:
(i) unity
(ii) zero
(iii) < 0
(iv) different for each element
Solution : The enthalpy of all elements in their standard state is zero. Therefore, alternative (ii) is correct
4. ΔU ⊖ of combustion of methane is −X kJ mol −1 . The value of ΔH⊖ is
(i)=ΔU ⊖
(ii) >ΔU ⊖
(iii) <=ΔU ⊖
(iv) = 0
Solution :
SinceΔH θ =ΔU θ +Δn g RT and ΔU θ =−Xkmol −1
ΔH θ =(−X)+Δn g RT
⇒△H θ <ΔU θ
Therefore, alternative (iii) is correct.
5. The enthalpy of combustion of methane, graphite and dihydrogen at 298 K are, –890.3kJmol −1 −393.5kJmol −1 , and −285.8kJmol −1
respectively. Enthalpy of formation of CH 4 will be
(i)−74.8kJmol −1
(ii)−52.27kJmol −1
(iii)+74.8kJmol −1
(iv)+52.26kJmol −1
Solution :
According to the question,
(i)CH 4 (g)+2O 2 (g)⟶CO 2 (z)+2H 2 O(g)ΔH=−890.3kJmol −1
(ii)C(x)+O 2 (y)⟶CO 2 (g)ΔH=−393.5kJmol −1
(iii)2H 2 (g)+O 2 (z)⟶2H 2 O(g)
ΔH=−285.8kJmol −1
Thus, the desired equation is the one that represents the formation of CH 4 (g) i.e..,
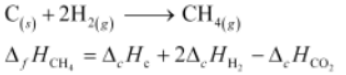
= [-395.5 + 2(-285.8) - (-890.3)] kJ Mol -1
= -74.8 kJ Mol -1
∴ Enthalpy of formation of CH 4 (g)=−74.8kJmol −1 Hence, alternative (i) is correct.
Read More: NCERT Solutions For Class 11 Chemistry Chapter 1
6. A reaction, A + B → C + D + q is found to have a positive entropy change. The reaction will be
(i) possible at high temperature
(ii) possible only at low temperature
(iii) not possible at any temperature
(iv) possible at any temperature
Solution :
For a reaction to be spontaneous, ΔG should be negative.
ΔG = ΔH – TΔS
According to the question, for the given reaction,
ΔS = positive
ΔH = negative (since heat is evolved)
⇒ ΔG = negative
Therefore, the reaction is spontaneous at any temperature.
Hence, alternative (iv) is correct.
7. In a process, 701 J of heat is absorbed by a system and 394 J of work is done by the system. What is the change in internal energy for the process?
Solution :
According to the first law of thermodynamics,
ΔU = q + W (i)
Where,
ΔU = change in internal energy for a process
q = heat
W = work
Given,
q = + 701 J (Since heat is absorbed)
W = –394 J (Since work is done by the system)
Substituting the values in expression (i), we get
ΔU = 701 J + (–394 J)
ΔU = 307 J
Hence, the change in internal energy for the given process is 307 J.
8. The reaction of cyanamide,NH 2 CN(s) with dioxygen was carried out in a bomb calorimeter, and ∆U was found to be –742.7kJmol −1 at 298 K. Calculate enthalpy change for the reaction at 298 K.
NH 2 CN(g) + 3/2O 2 (g)→N 2 (g)+CO 2 (g)+H 2 O(l)
Solution :
Enthalpy change for a reaction (ΔH) is given by the expression,
ΔH = ΔU + Δn g RT
Where,
ΔU = change in internal energy
Δn g = change in number of moles
For the given reaction,
Δng = ∑n g (products) – ∑n g (reactants)
= (2 – 1.5) moles
Δn g = 0.5 moles
And,
ΔU = –742.7 kJ mol –1
T = 298 K
R = 8.314 × 10 –3 kJ mol –1 K –1
Substituting the values in the expression of ΔH:
ΔH = (–742.7 kJ mol –1 ) + (0.5 mol) (298 K) (8.314 × 10 –3 kJ mol –1 K –1 )
= –742.7 + 1.2
ΔH = –741.5 kJ mol –1
9. Calculate the number of kJ of heat necessary to raise the temperature of 60.0 g of aluminium from 35°C to 55°C. Molar heat capacity of Al is 24 Jmol −1 K −1 .
Solution :
From the expression of heat (q) q=m⋅ c. ΔT
Where,
c= molar heat capacity
m= mass of substance
ΔT= change in temperature
Substituting the values in the expression of q:
q=(60/27mol)(24Jmol −1 K −1 )(20K)
q=1066.7J
q=1.07k
10. Calculate the enthalpy change on freezing of 1.0 mol of water at10.0°C to ice at
–10.0°C.ΔfusH=6.03kJmol−1 at 0 ∘ C
C p [H 2 O(I)]=75.3Jmol −1 K−1
C ρ [H 2 O(s)]=36.8Jmol −1 K −1
Solution :
Total enthalpy change involved in the transformation is the of the following changes:
(a) Energy change involved in the transformation of 1 mol of water at 10 ∘ C to 1 mol of water at 0 C.
(b) Energy change involved in the transformation of 1 mol of water at 0∘ to 1 mol of ice at 0 ∘ C
(c) Energy change involved in the transformation of 1 mol of ice at0∘C to 1 mol of ice at−10 ∘ C.
ΔH= C p [ H 2 OCl]ΔT+ ΔH fivering + C ρ [H 2 O (s) ]ΔT
=(75.3] mol −1 K −1 )(0−10)K + (−6.03 × 10 3 Jmol −1 )+(36.8] mol −1 K −1 )(−10 −0)K
=−7533 mol −1 − 6030Jmol −1 − 368Jmol −1
=−7151J mol −1
=−7.151kJmol −1
Hence, the enthalpy change involved in the transformation is−7.151kJmol −1 .
Read More: NCERT Solutions for Class 11 Chemistry Chapter 2
11. Enthalpy of combustion of carbon to CO 2 is –393.5−7.151kJmol −1 . Calculate the heat released upon formation of 35.2 g ofCO 2 from carbon and dioxygen gas.
Solution :
Formation of CO 2 from carbon and dioxygen gas can be represented as:
C(s) + O 2 (g)⟶CO 2 (g)
Δ f H=−393.5kJmol −1
(1 mole =44g) Heat released on formation of 44gCO 2 =−393.5kJmol−1
∴ Heat released on formation of 35.2gCO 2
=−314.8kJmol −1
12. Enthalpies of formation of CO(g), CO 2 (g), N 2 O(g) and N 2 O 4 (g) are −110,−393,81 and 9.7kJmol −1 respectively.
Find the value of ∆H for the reaction:
N 2 O 4 (g)+3CO(g)→N 2 O(g+3CO 2 (g]
Solution :
Δ r H for a reaction is defined as the difference between ΔH value of products and ΔH value of reactants.
Δ,H=∑Δ,H( products )−∑Δ f H( reactants )
For the given reaction,
N 2 O 4(g) + 3CO (g) ⟶ N 2 O (g) + 3CO 2 (g)
Δ r H=[ {ΔfH(N 2 O)+3ΔJH(CO 2 )}−{ΔfH (N 2 O 4 ) + 3ΔjH(CO)} ]
Substituting the values ofΔH for N 2 O,CO 2 ,N 2 O 4 , and CO
From the question, we get:
Δ r H=[ { 81kJmol −1 + 3(−393)kJmol −1 } − {9.7kJmol −1 +3(−110) kJmol −1 }]
Δ r H=−7777kJmol −1
Hence, the value ofΔ r H
for the reaction is −777.7 kJmol −1 .
13. Given
N 2 (g) + 3H 2 (g) ⟶ 2NH 3 (y);
Δ r Hθ=−92.4kJmol −1
What is the standard enthalpy of formation ofNH 3 gas?
Solution : Standard of formation of a compound is the charge in enthalpy that takes place during the formation of 1 mole Of a substance in its standard form from its constituent elements in their standard state.
Re-writing the given equation for 1 mole of NH 3(g).
1/2N 2 (g)+3/2H 2 (g)⟶NH 3(g)
∴ Standard enthalpy of formation of NH 3(g)
=1/2 Δ r Hθ = 1/2(−92.4 kJmol −1 )= −46.2kJmol −1
14. Calculate the standard enthalpy of formation ofCH 3 OH(l) from the following data:
CH 3 OH(l)+3/2O 2 (g]→CO 2 (g)+2H 2 O(l): Δ,H∘=−726kJmol −1
C(graphite) +O 2 (g)→CO2(g]: ΔeH=−393kJmol −1
H 2 (g)+1/2O 2 (g)→H 2 O(1); Δ,H=−286kJmol −1
Solution :
The reaction that takes place during the formation ofCH 3 OH(l) can be written as:
C(s) + 2H 2 O(g) + 1/2O 2 (G), ⟶ CH 3 OH( η) (1)
The reaction (I) can be obtained from the given reactions by following the algebraic calculations as:
Equation (ii) + 2 × equation (iii) – equation (i)
Δ f Hθ [CH 3 OH(l)] = ΔcH θ + 2Δ f H θ [H 2 O(l)] – Δ r H θ
= (–393 kJ mol –1 ) + 2(–286 kJ mol –1 ) – (–726 kJ mol –1 )
= (–393 – 572 + 726) kJ mol –1
Δ f H θ [CH 3 OH(l)] = –239 kJ mol –1
15. Calculate the enthalpy change for the process CCl 4 (g)→C(g)+4Cl(g) and calculated bond enthalpy of C−Cl in CCl 4 (g)
Δ va pH θ (CC|4) = 30.5kJmol −1 Δ f H θ (CCl4) =−135.5kJmol−1
Δ a H θ (C) = 715.0kJmol −1 , where Δ a H θ is enthalpy of atomisation
Δ 2 H θ (Cl 2 ) = 242kJmol −1
Solution :
The chemical equations implying to the given values of enthalpies” are:
(1) CCl 4(l) à CCl 4(g) ; ΔvapH Θ = 30.5 kJmol −1
(2) C (s) à C (g) ΔaH Θ = 715 kJmol −1
(3) Cl2 (g) à 2Cl (g) ; Δ a H Θ = 242 kJmol −1
(4) C (g) + 4Cl (g) à CCl 4(g) ; ΔfH Θ
= -135.5 kJmol −1 ΔH for the process CCl 4(g) à C (g) + 4Cl (g) can be measured as:
ΔH=Δ a H Θ (C) + 2ΔaH Θ (Cl 2 ) – Δ vap H Θ –ΔfH
= (715kJmol −1 ) + 2(kJmol −1 ) – (30.5kJmol −1 ) – (-135.5kJmol −1 )
Therefore, H= 1304kJmol −1
The value of bond enthalpy for C-Cl in CCl 4 (g)
= 1304/4kJmol −1
= 326 kJmol −1
16. For an isolated system, ∆U = 0, what will be ∆S ?
Solution :
ΔS will be positive i.e., greater than zero
Since ΔU = 0, ΔS will be positive and the reaction will be spontaneous.
Read More: NCERT Solutions For Class 11 Chemistry Chapter 3
17. For the reaction at 298 K,
2A + B → C
∆H = 400kJmol −1
and ∆S = 0.2kJmol −1
At what temperature will the reaction become spontaneous considering ∆H and ∆S to be constant over the temperature range.
Solution :
From the expression
ΔG= ΔH−TΔS
Assuming the reaction at equilibrium,δ
T for the reaction would be:
T=(ΔH−ΔG)1/ΔS=ΔH/ΔS(ΔG=0 at equilibrium)
=400kJmol −1 0.2 kJK −1 mol −1 T=2000K
For the reaction to be spontaneous,ΔG must be negative. Hence, for the given reaction to be spontaneous, T should be greater than 2000 K.
18. For the reaction,2Cl(g)→Cl 2 (g) , what are the signs of ∆H and ∆S ?
Solution :
∆H and ∆S are negative
The given reaction represents the formation of chlorine molecule from chlorine atoms. Here, bond formation is taking place. Therefore, energy Is being released. Hence ∆H is negative.
Also, two moles of atoms have more randomness than one mole of a molecule. Since spontaneity is decreased, ∆S is negative for the given reaction.
19. For the reaction
2A(g)+B(g)→2D(g)ΔUe=−10.5kJ and ΔS∘=−44.1JK −1
Calculate ΔG ⊖ for the reaction, and predict whether the reaction may occur spontaneously
Solution :
For the given reaction,
2A(g)+B(g)→2D(g)Δηg=2−(3)=−1 mole
Substituting the value of ΔU θ
in the expression of ΔH:
ΔH θ =ΔU θ +Δn g RT
=(−10.5kJ)−(−1)(8.314×10 −3 kJK−1 mol −1 ) (298K) = −10.5kJ −2 .48kJΔH ⊖ =−12.98kJ
Substituting the values ofΔH ⊖ and ΔS ⊖ in the expression of ΔG ⊖
ΔG θ = △H θ −TΔS θ
=−12.98kJ − ( 298K) (−44.1JK −1 ) = −12.98kJ + 13.14 kJ ΔG ⊖ =+0.16kJ
SinceΔG θ for the reaction is positive, the reaction will not occur spontaneously.
20. The equilibrium constant for a reaction is 10. What will be the value of ∆G ⊖ ? R = 8.314JK −1 mol −1
T = 300 K.
Solution :
From the expression, ΔG θ = −2.303 RT logk eq
ΔG θ for the reaction,
=(2.303) (8.314JK −1 mol −1 ) (300K) log10=−5744.14Jmol −1
=−5.744kkmol −1
21. Comment on the thermodynamic stability of NO(g) , given
12N 2(g) +12O 2(g) →NO (g) ;
Δ r H ⊖ = 90kJmol −1 NO (g) + 12O 2(g) →NO 2 (g):
Δ r H e = −74kJmol −1
Solution :
The positive value of Δ r H indicates that heat is absorbed during the formation of NO(g), j. This means that NO(g) has higher than the reactants(N2 and O2) .
Hence, NO (g) is unstable. The negative value o f Δ r H
H indicates that heat is evolved during the formation ofNO 2(g) from NO (g) and O 2(g)
. The product,NO 2(g) is stabilized with minimum energy.
Hence, unstableNO (g) changes to unstableNO 2(g).
22. Calculate the entropy change in surroundings when 1.00 mol ofH 2 O(l) is formed under standard conditions.ΔH θ =−286kJ mol −1
Solution :
It is given that 286 kJmol −1 of heat is evolved the formation of 1 mol ofH 2 O(l).
Thus, an equal amount of heat will be absorbed by the surroundings.
q surr = +286 kJmol −1
Entropy change(ΔS surr ) for the surroundings = q surr / 7
=286kJmol −1 / 298k
∴ΔS surt =959.73 Jmol −1 K −1
Read More: NCERT Solutions for Class 11 Chemistry Chapter 4
Understanding Intermolecular Forces and Thermal Energy
The behavior of any substance is determined by a constant "tug-of-war" between intermolecular forces and thermal energy. According to the class 11 chemistry chapter 6 ncert materials, intermolecular forces are the attractive and repulsive forces between interacting particles. These are distinct from intramolecular forces, such as the covalent bonds that hold a single molecule together.
In this chapter, we focus heavily on Van der Waals forces, which include dispersion forces (London forces), dipole-dipole forces, and dipole-induced dipole forces. A key takeaway for students is how these forces dictate physical properties like boiling points and melting points. For instance, London forces are the weakest and occur between non-polar molecules due to temporary dipoles. On the other hand, dipole-dipole forces act between molecules with permanent dipoles.
The Unique Framing: The "State Equilibrium" Perspective
While most guides treat gases, liquids, and solids as static categories, a more helpful way to view them is through "State Equilibrium." Imagine a scale where Intermolecular Attraction is on one side and Thermal Energy is on the other. In gases, Thermal Energy heavily outweighs Attraction, leading to chaos. In solids, Attraction wins. Understanding this balance helps you predict how a substance will react when you manipulate temperature or pressure, rather than just memorizing the gas laws.
Mastering the Gas Laws in Class 11 Chemistry Chapter 6 NCERT Solutions
The heart of this chapter lies in the measurable properties of gases: mass, volume, pressure, and temperature. The class 11 chemistry chapter 6 ncert solutions provide a step-by-step breakdown of the fundamental laws that govern these variables.
-
Boyle’s Law (Pressure-Volume Relationship): At a constant temperature, the pressure of a fixed amount of gas varies inversely with its volume (P \propto 1/V). This means if you halve the volume, the pressure doubles.
-
Charles’s Law (Temperature-Volume Relationship): At constant pressure, the volume of a gas is directly proportional to its absolute temperature (V \propto T). This explains why gases expand when heated.
-
Gay-Lussac’s Law (Pressure-Temperature Relationship): At constant volume, pressure is directly proportional to temperature.
-
Avogadro’s Law: This states that equal volumes of all gases under the same conditions of temperature and pressure contain an equal number of molecules.
When you combine these, you arrive at the Ideal Gas Equation: PV = nRT. Mastering the class 11 chemistry chapter 6 ncert question answer sections involves practicing the conversion of units (like converting Celsius to Kelvin) to ensure the R-value (gas constant) remains accurate in your calculations.
Check Out: Class 11th Question Banks
Kinetic Molecular Theory and Deviation from Ideal Behavior
Why do we distinguish between "Ideal" and "Real" gases? The Kinetic Molecular Theory provides a theoretical model for an ideal gas, assuming that gas particles have no volume and no attractive forces between them. However, the class 11 chemistry chapter 6 ncert book pdf clarifies that real gases only behave ideally under specific conditions: low pressure and high temperature.
At high pressure or low temperature, the intermolecular forces become significant, and the volume of the particles themselves can no longer be ignored. This leads to the use of the Van der Waals equation, which introduces constants to account for these "real-world" factors. When studying for exams, focus on the Compressibility Factor (Z). If Z = 1, the gas is ideal. If Z is not 1, the gas shows deviation. Understanding this transition is vital for students who will later study the energy changes in class 11 chemistry chapter 6 ncert solutions thermodynamics.
The Liquid State: Vapor Pressure, Viscosity, and Surface Tension
While gases occupy much of the chapter, the liquid state provides fascinating insights into fluid dynamics. The class 11 chemistry chapter 6 ncert content highlights three main properties:
-
Vapor Pressure: In a closed vessel, the pressure exerted by the vapors in equilibrium with the liquid at a specific temperature. As temperature increases, vapor pressure increases.
-
Surface Tension: This is the force acting per unit length perpendicular to a line drawn on the surface of the liquid. It is why droplets are spherical—nature seeks the minimum surface area.
-
Viscosity: This is a measure of a liquid's resistance to flow. Stronger intermolecular forces lead to higher viscosity. For example, honey has higher viscosity than water because its internal "friction" or resistance between layers is much greater.
Practicing the class 11 chemistry chapter 6 ncert question answer sets related to these properties will help you understand why boiling points vary with altitude, as external pressure influences when vapor pressure equals atmospheric pressure.
PW CBSE Class 11 Study Material
Complete CBSE Syllabus Coverage
PW CBSE Class 11 Study Material is prepared according to the latest CBSE and NCERT syllabus. It covers all major subjects such as Physics, Chemistry, Maths, and Biology in a structured manner.
Simple and Concept-Based Explanations
All topics are explained in clear and simple language with examples and diagrams. This helps students understand concepts easily and build strong fundamentals.
Chapter-Wise Notes
Well-organised chapter-wise notes allow students to study systematically and revise important topics quickly.
Practice Questions and Solved Examples
The study material includes solved examples and practice questions to improve problem-solving skills and exam readiness.
Sample Papers for Exam Practice
Latest sample papers based on the CBSE pattern help students practise effectively, manage time better, and gain exam confidence.
Strong Foundation for Future Exams
With clear concepts and regular practice, this study material supports preparation for Class 12 and competitive exams.
Also Check: CBSE Question Bank Class 11 Chemistry
NCERT Solutions for Class 11 Chemistry chapter 6 FAQs
Q1: What is the most important formula in Class 11 Chemistry chapter 6?
The Ideal Gas Equation, PV = nRT, is the most central formula. It combines Boyle's, Charles's, and Avogadro's laws and is used extensively in both the class 11 chemistry chapter 6 ncert question answer sets and future chapters like thermodynamics.
Q2: How do real gases differ from ideal gases?
Ideal gases follow the gas laws at all temperatures and pressures, assuming no intermolecular attractions. Real gases deviate from this behavior, especially at high pressure and low temperature, which is why the Van der Waals equation is used.
Q3: What is the significance of the 'Compressibility Factor' (Z)?
Z is the ratio of PV to nRT. It tells you how much a real gas deviates from ideal behavior. For an ideal gas, Z is always 1.
Q4: How does surface tension change with temperature?
As temperature increases, the kinetic energy of the molecules increases, which decreases the effectiveness of intermolecular attractions. Consequently, surface tension decreases as temperature rises.
Q5: Can I find the Class 11 Chemistry chapter 6 NCERT Book PDF online for free?
Yes, the official NCERT website provides the class 11 chemistry chapter 6 ncert book pdf for free. Using this alongside PW study materials ensures you have the official curriculum and the expert strategy needed to score well.










