NCERT Solutions for Class 12 Chemistry Chapter 1 The Solid State

NCERT Solutions for Class 12 Chemistry Chapter 1 – The Solid State explain the basic ideas of how solids are formed and how their particles are arranged. In simple words, this chapter helps students understand the difference between crystalline solids (where particles are arranged in a fixed pattern) and amorphous solids (where particles are not properly arranged).
It also explains important topics like unit cells, crystal lattices, and different types of crystal structures such as cubic and hexagonal. Students learn how particles are packed in solids and how to calculate things like density and packing efficiency in an easy step-by-step way through NCERT solutions.
The chapter also includes defects in solids, which means small imperfections in the structure of solids. These defects are important because they affect the properties of materials used in real life, like metals and semiconductors.
NCERT solutions make this chapter easy to understand by giving clear answers, simple explanations, and solved numerical problems. It helps students prepare well for exams and build strong basic concepts in chemistry.
Check Out: CBSE Class 12 Books
NCERT Solutions for Class 12 Chemistry Chapter 1 Overview
NCERT Solutions for Class 12 Chemistry Chapter 1 – The Solid State give a simple and clear overview of how solids are structured and behave. This chapter explains the difference between crystalline and amorphous solids, along with basic ideas like unit cells, crystal lattices, and packing of particles. It also includes easy explanations of important numerical topics like density and packing efficiency. These solutions help students understand concepts in a step-by-step way, making exam preparation easier and more effective.
NCERT Solutions for Class 12 Chemistry Chapter 1
The answers and explanations for the exercise questions in the textbook are below. These Chemistry Class 12 Chapter 1 NCERT solutions are made to meet the rules for grading used in board exams.
Question 1. Name the parameters that characterize a unit cell.
Solution : The six parameters that characterise a unit cell are as follows. (i) Its dimensions along the three edges, a, b, and c These edges may or may not be equal. (ii) Angles between the edges These are the angle ∝ (between edges b and c), β (between edges a and c), and γ (between edges a and b).
Question 2. Distinguish between (i)Hexagonal and monoclinic unit cells (ii) Face-centred and end-centred unit cells.
Solution : (i) Hexagonal unit cell For a hexagonal unit cell, Monoclinic unit cell For a monoclinic cell,
Monoclinic unit cell For a monoclinic cell,
(ii) Face-centred unit cell In a face-centred unit cell, the constituent particles are present at the corners and one at the centre of each face. End-centred unit cell An end-centred unit cell contains particles at the corners and one at the centre of any two opposite faces.
Question 3. Explain how much portion of an atom located at (i) corner and (ii) body-centre of a cubic unit cell is part of its neighbouring unit cell.
Solution : (i)An atom located at the corner of a cubic unit cell is shared by eight adjacent unit cells.
Therefore, 1/8 th portion of the atom is shared by one unit cell. (ii)An atom located at the body centre of a cubic unit cell is not shared by its neighbouring unit cell. Therefore, the atom belongs only to the unit cell in which it is present i.e., its contribution to the unit cell is 1.
Question 4. What is the two dimensional coordination number of a molecule in square close packed layer?
Solution : In square close-packed layer, a molecule is in contact with four of its neighbours. Therefore, the two-dimensional coordination number of a molecule in square close-packed layer is 4.
Question 5. A compound forms hexagonal close-packed structure.
What is the total number of voids in 0.5 mol of it? How many of these are tetrahedral voids?
Solution : Number of close-packed particles = 0.5 × 6.022 × 10 23 = 3.011 × 10 23 Therefore, number of octahedral voids = 3.011 × 10 23 And, number of tetrahedral voids = 2 × 3.011 × 10 23 = 6.022 ×10 23 Therefore, total number of voids = 3.011 × 10 23 + 6.022 × 10 23 = 9.033 × 10 23
Read More: NCERT Solutions for Class 12 Chemistry Chapter 2
Question 6. A compound is formed by two elements M and N. The element N forms ccp and atoms of M occupy 1/3 rd of tetrahedral voids.
What is the formula of the compound?
Solution : The ccp lattice is formed by the atoms of the element N. Here, the number of tetrahedral voids generated is equal to twice the number of atoms of the element N.
According to the question, the atoms of element M occupy 1/3 rd of the tetrahedral voids. Therefore, the number of atoms of M is equal to of the number of atoms of N. Therefore, ratio of the number of atoms of M to that of N is M: N
of the number of atoms of N. Therefore, ratio of the number of atoms of M to that of N is M: N = 2 : 3 Thus, the formula of the compound is M 2 N 3 .
= 2 : 3 Thus, the formula of the compound is M 2 N 3 .
Solution : Hexagonal close-packed lattice has the highest packing efficiency of 74%. The packing efficiencies of simple cubic and body-centred cubic lattices are 52.4% and 68% respectively.
Question 8. An element with molar mass 2.7 × 10 -2 kg mol -1 forms a cubic unit cell with edge length 405 pm. If its density is 2.7 × 103 kg m −3 , what is the nature of the cubic unit cell?
Solution : It is given that density of the element, d = 2.7 × 10 3 kg m −3 Molar mass, M = 2.7 × 10 −2 kg mol −1 Edge length, a = 405 pm = 405 × 10 −12 m = 4.05 × 10 −10 m It is known that, Avogadro’s number, N A = 6.022 × 1023 mol −1 Applying the relation,

This implies that four atoms of the element are present per unit cell. Hence, the unit cell is face-centred cubic (fcc) or cubic close-packed (ccp).
Question 9. What type of defect can arise when a solid is heated? Which physical property is affected by it and in what way?
Solution : When a solid is heated, vacancy defect can arise. A solid crystal is said to have vacancy defect when some of the lattice sites are vacant. Vacancy defect leads to a decrease in the density of the solid.
Question 10. What type of stoichiometric defect is shown by: (i) ZnS (ii) AgBr
Solution : (i) ZnS shows Frenkel defect. (ii) AgBr shows Frenkel defect as well as Schottky defect.
Question 11. Explain how vacancies are introduced in an ionic solid when a cation of higher valence is added as an impurity in it.
Solution : When a cation of higher valence is added to an ionic solid as an impurity to it, the cation of higher valence replaces more than one cation of lower valence so as to keep the crystal electrically neutral. As a result, some sites become vacant. For example, when Sr 2+ is added to NaCl, each Sr 2+ ion replaces two Na + ions. However, one Sr 2+ ion occupies the site of one Na + ion and the other site remains vacant. Hence, vacancies are introduced.
Question 12. Ionic solids, which have anionic vacancies due to metal excess defect, develop colour. Explain with the help of a suitable example.
Solution : The colour develops because of the presence of electrons in the anionic sites. These electrons absorb energy from the visible part of radiation and get excited. For example, when crystals of NaCl are heated in an atmosphere of sodium vapours, the sodium atoms get deposited on the surface of the crystal and the chloride ions from the crystal diffuse to the surface to form NaCl with the deposited Na atoms. During this process, the Na atoms on the surface lose electrons to form Na + ions and the released electrons diffuse into the crystal to occupy the vacant anionic sites. These electrons get excited by absorbing energy from the visible light and impart yellow colour to the crystals.
Question 13. A group 14 element is to be converted into n-type semiconductor by doping it with a suitable impurity. To which group should this impurity belong?
Solution : An n-type semiconductor conducts because of the presence of extra electrons. Therefore, a group 14 element can be converted to n-type semiconductor by doping it with a group 15 element.
Question 14. What type of substances would make better permanent magnets, ferromagnetic or ferrimagnetic. Justify your answer.
Solution : Ferromagnetic substances would make better permanent magnets. In solid state, the metal ions of ferromagnetic substances are grouped together into small regions. These regions are called domains and each domain acts as a tiny magnet. In an unmagnetised piece of a ferromagnetic substance, the domains are randomly oriented. As a result, the magnetic moments of the domains get cancelled. However, when the substance is placed in a magnetic field, all the domains get oriented in the direction of the magnetic field and a strong magnetic effect is produced. The ordering of the domains persists even after the removal of the magnetic field. Thus, the ferromagnetic substance becomes a permanent magnet.
Question 15. Why are solids rigid?
Solution : The intermolecular forces of attraction that are present in solids are very strong. The constituent particles of solids cannot move from their positions i.e., they have fixed positions. However, they can oscillate about their mean positions. This is the reason solids are rigid.
 p-type semiconductor: The semiconductor whose increased in conductivity is a result of electron hole is called a p-type semiconductor. When a crystal of group 14 elements such as Si or Ge is doped with a group 13 element such as B, Al, or Ga (which contains only three valence electrons), a p-type of semiconductor is generated. When a crystal of Si is doped with B, the three electrons of B are used in the formation of three covalent bonds and an electron hole is created. An electron from the neighbouring atom can come and fill this electron hole, but in doing so, it would leave an electron hole at its original position. The process appears as if the electron hole has moved in the direction opposite to that of the electron that filled it. Therefore, when an electric field is applied, electrons will move toward the positively-charged plate through electron holes. However, it will appear as if the electron holes are positively-charged and are moving toward the negatively- charged plate.
p-type semiconductor: The semiconductor whose increased in conductivity is a result of electron hole is called a p-type semiconductor. When a crystal of group 14 elements such as Si or Ge is doped with a group 13 element such as B, Al, or Ga (which contains only three valence electrons), a p-type of semiconductor is generated. When a crystal of Si is doped with B, the three electrons of B are used in the formation of three covalent bonds and an electron hole is created. An electron from the neighbouring atom can come and fill this electron hole, but in doing so, it would leave an electron hole at its original position. The process appears as if the electron hole has moved in the direction opposite to that of the electron that filled it. Therefore, when an electric field is applied, electrons will move toward the positively-charged plate through electron holes. However, it will appear as if the electron holes are positively-charged and are moving toward the negatively- charged plate.

 On the other hand, in the case of an insulator, the valence band is fully- filled and there is a large gap between the valence band and the conduction band. (ii) In the case of a conductor, the valence band is partially-filled or it overlaps with a higher energy, unoccupied conduction band. So, the electrons can flow easily under an applied electric field.
On the other hand, in the case of an insulator, the valence band is fully- filled and there is a large gap between the valence band and the conduction band. (ii) In the case of a conductor, the valence band is partially-filled or it overlaps with a higher energy, unoccupied conduction band. So, the electrons can flow easily under an applied electric field.
 On the other hand, the valence band of a semiconductor is filled and there is a small gap between the valence band and the next higher conduction band. Therefore, some electrons can jump from the valence band to the conduction band and conduct electricity.
On the other hand, the valence band of a semiconductor is filled and there is a small gap between the valence band and the next higher conduction band. Therefore, some electrons can jump from the valence band to the conduction band and conduct electricity. (ii) Frenkel defect: Ionic solids containing large differences in the sizes of ions show this type of defect. When the smaller ion (usually cation) is dislocated from its normal site to an interstitial site, Frenkel defect is created. It creates a vacancy defect as well as an interstitial defect. Frenkel defect is also known as dislocation defect. Ionic solids such as AgCl, AgBr, AgI, and ZnS show this type of defect.
(ii) Frenkel defect: Ionic solids containing large differences in the sizes of ions show this type of defect. When the smaller ion (usually cation) is dislocated from its normal site to an interstitial site, Frenkel defect is created. It creates a vacancy defect as well as an interstitial defect. Frenkel defect is also known as dislocation defect. Ionic solids such as AgCl, AgBr, AgI, and ZnS show this type of defect. (iii) Interstitials: Interstitial defect is shown by non-ionic solids. This type of defect is created when some constituent particles (atoms or molecules) occupy an interstitial site of the crystal. The density of a substance increases because of this defect.
(iii) Interstitials: Interstitial defect is shown by non-ionic solids. This type of defect is created when some constituent particles (atoms or molecules) occupy an interstitial site of the crystal. The density of a substance increases because of this defect. (iv) F-centres: When the anionic sites of a crystal are occupied by unpaired electrons, the ionic sites are called F-centres. These unpaired electrons impart colour to the crystals. For example, when crystals of NaCl are heated in an atmosphere of sodium vapour, the sodium atoms are deposited on the surface of the crystal. The Cl ions diffuse from the crystal to its surface and combine with Na atoms, forming NaCl. During this process, the Na atoms on the surface of the crystal lose electrons. These released electrons diffuse into the crystal and occupy the vacant anionic sites, creating F-centres.
(iv) F-centres: When the anionic sites of a crystal are occupied by unpaired electrons, the ionic sites are called F-centres. These unpaired electrons impart colour to the crystals. For example, when crystals of NaCl are heated in an atmosphere of sodium vapour, the sodium atoms are deposited on the surface of the crystal. The Cl ions diffuse from the crystal to its surface and combine with Na atoms, forming NaCl. During this process, the Na atoms on the surface of the crystal lose electrons. These released electrons diffuse into the crystal and occupy the vacant anionic sites, creating F-centres.
 = 2.27 × 10 22
= 2.27 × 10 22 mol of SrCl 2 = 10 −5 mol of SrCl 2 Cation vacancies produced by one Sr 2+ ion = 1
mol of SrCl 2 = 10 −5 mol of SrCl 2 Cation vacancies produced by one Sr 2+ ion = 1 Hence, the concentration of cation vacancies created by SrCl 2 is 6.022 × 10 8 per mol of NaCl.
Hence, the concentration of cation vacancies created by SrCl 2 is 6.022 × 10 8 per mol of NaCl. Schematic alignment of magnetic moments in ferromagnetic substances (ii) Paramagnetism: The substances that are attracted by a magnetic field are called paramagnetic substances. Some examples of paramagnetic substances are O 2 , Cu 2t , Fe 3t , and Cr 3t . Paramagnetic substances get magnetised in a magnetic field in the same direction, but lose magnetism when the magnetic field is removed. To undergo paramagnetism, a substance must have one or more unpaired electrons. This is because the unpaired electrons are attracted by a magnetic field, thereby causing paramagnetism. (iii) Ferrimagnetism: The substances in which the magnetic moments of the domains are aligned in parallel and anti-parallel directions, in unequal numbers, are said to have ferrimagnetism. Examples include Fe 3 O 4 (magnetite), ferrites such as MgFe 2 O 4 and ZnFe 2 O 4 . Ferrimagnetic substances are weakly attracted by a magnetic field as compared to ferromagnetic substances. On heating, these substances become paramagnetic.
Schematic alignment of magnetic moments in ferromagnetic substances (ii) Paramagnetism: The substances that are attracted by a magnetic field are called paramagnetic substances. Some examples of paramagnetic substances are O 2 , Cu 2t , Fe 3t , and Cr 3t . Paramagnetic substances get magnetised in a magnetic field in the same direction, but lose magnetism when the magnetic field is removed. To undergo paramagnetism, a substance must have one or more unpaired electrons. This is because the unpaired electrons are attracted by a magnetic field, thereby causing paramagnetism. (iii) Ferrimagnetism: The substances in which the magnetic moments of the domains are aligned in parallel and anti-parallel directions, in unequal numbers, are said to have ferrimagnetism. Examples include Fe 3 O 4 (magnetite), ferrites such as MgFe 2 O 4 and ZnFe 2 O 4 . Ferrimagnetic substances are weakly attracted by a magnetic field as compared to ferromagnetic substances. On heating, these substances become paramagnetic. Schematic alignment of magnetic moments in ferrimagnetic substances (iv) Antiferromagnetism: Antiferromagnetic substanceshave domain structures similar to ferromagnetic substances, but are oppositely-oriented. The oppositely-oriented domains cancel out each other’s magnetic moments.
Schematic alignment of magnetic moments in ferrimagnetic substances (iv) Antiferromagnetism: Antiferromagnetic substanceshave domain structures similar to ferromagnetic substances, but are oppositely-oriented. The oppositely-oriented domains cancel out each other’s magnetic moments. Schematic alignment of magnetic moments in antiferromagnetic substances (v) 12-16 and 13-15 group compounds: The 12-16 group compounds are prepared by combining group 12 and group 16 elements and the 13-15 group compounds are prepared by combining group 13 and group15 elements. These compounds are prepared to stimulate average valence of four as in Ge or Si. Indium (III) antimonide (IrSb), aluminium phosphide (AlP), and gallium arsenide (GaAS) are typical compounds of groups 13-15. GaAs semiconductors have a very fast response time and have revolutionised the designing of semiconductor devices. Examples of group 12-16 compounds include zinc sulphide (ZnS), cadmium sulphide (CdS), cadmium selenide (CdSe), and mercury (II) telluride (HgTe). The bonds in these compounds are not perfectly covalent. The ionic character of the bonds depends on the electronegativities of the two elements.
Schematic alignment of magnetic moments in antiferromagnetic substances (v) 12-16 and 13-15 group compounds: The 12-16 group compounds are prepared by combining group 12 and group 16 elements and the 13-15 group compounds are prepared by combining group 13 and group15 elements. These compounds are prepared to stimulate average valence of four as in Ge or Si. Indium (III) antimonide (IrSb), aluminium phosphide (AlP), and gallium arsenide (GaAS) are typical compounds of groups 13-15. GaAs semiconductors have a very fast response time and have revolutionised the designing of semiconductor devices. Examples of group 12-16 compounds include zinc sulphide (ZnS), cadmium sulphide (CdS), cadmium selenide (CdSe), and mercury (II) telluride (HgTe). The bonds in these compounds are not perfectly covalent. The ionic character of the bonds depends on the electronegativities of the two elements.  ⇒ d =zm / a 3----------------------------(1) [Since mass of the unit cell = Number of atoms in the unit cell × mass of one atom] [Volume of the unit cell = (Edge length of the cubic unit cell) 3 ] From equation (i), we have: m = da 3 /z -----------------------------------------(ii) Now, mass of one atom of metal (m)
⇒ d =zm / a 3----------------------------(1) [Since mass of the unit cell = Number of atoms in the unit cell × mass of one atom] [Volume of the unit cell = (Edge length of the cubic unit cell) 3 ] From equation (i), we have: m = da 3 /z -----------------------------------------(ii) Now, mass of one atom of metal (m) Therefore,
Therefore, If the edge lengths are different (say a, b and c), then equation (ii) becomes: M = d(abc)NAz (iv) From equations (iii) and (iv), we can determine the atomic mass of the unknown metal.
If the edge lengths are different (say a, b and c), then equation (ii) becomes: M = d(abc)NAz (iv) From equations (iii) and (iv), we can determine the atomic mass of the unknown metal.
 Let ,the edge length = a Radius of each particle = r. Thus, a = 2r Volume of spheres = πr 3 (4/3) Volume of a cubic unit cell = a 3 = (2r) 3 = 8r 3 We know that the number of particles per unit cell is 1. Therefore, Packing efficiency = Volume of one particle/Volume of cubic unit cell = [ πr 3 (4/3) ] / 8r 3 = 0.524 or 52.4 % (b) Body-centred cubic:
Let ,the edge length = a Radius of each particle = r. Thus, a = 2r Volume of spheres = πr 3 (4/3) Volume of a cubic unit cell = a 3 = (2r) 3 = 8r 3 We know that the number of particles per unit cell is 1. Therefore, Packing efficiency = Volume of one particle/Volume of cubic unit cell = [ πr 3 (4/3) ] / 8r 3 = 0.524 or 52.4 % (b) Body-centred cubic: From ∆FED, we have: b 2 = 2a 2 b = ( 2a ) 1/2 Again, from ∆AFD, we have : c 2 = a 2 + b 2 => c 2 = a 2 + 2a 2 c 2 = 3a 2 => c = (3a) 1/2 Let the radius of the atom = r. Length of the body diagonal, c = 4r => (3a) 1/2 = 4r => a = 4r/ (3) 1/2 or , r = [ a (3) 1/2 ]/ 4 Volume of the cube, a 3 = [4r/ (3) 1/2 ] 3 A BCC lattice has 2 atoms. So, volume of the occupied cubic lattice = 2πr 3 (4/3) = πr 3 ( 8/3) Therefore, packing efficiency = [ πr 3 ( 8/3) ]/ [ { 4r/(3) 1/2 } 3 ] = 0.68 or 68% (iii) Face-centred cubic:
From ∆FED, we have: b 2 = 2a 2 b = ( 2a ) 1/2 Again, from ∆AFD, we have : c 2 = a 2 + b 2 => c 2 = a 2 + 2a 2 c 2 = 3a 2 => c = (3a) 1/2 Let the radius of the atom = r. Length of the body diagonal, c = 4r => (3a) 1/2 = 4r => a = 4r/ (3) 1/2 or , r = [ a (3) 1/2 ]/ 4 Volume of the cube, a 3 = [4r/ (3) 1/2 ] 3 A BCC lattice has 2 atoms. So, volume of the occupied cubic lattice = 2πr 3 (4/3) = πr 3 ( 8/3) Therefore, packing efficiency = [ πr 3 ( 8/3) ]/ [ { 4r/(3) 1/2 } 3 ] = 0.68 or 68% (iii) Face-centred cubic: Let the edge length of the unit cell = a let the radius of each sphere = r Thus, AC = 4r From the right angled triangle ABC, we have : AC = ( a 2 + a 2 ) 1/2 = a(2) 1/2
Let the edge length of the unit cell = a let the radius of each sphere = r Thus, AC = 4r From the right angled triangle ABC, we have : AC = ( a 2 + a 2 ) 1/2 = a(2) 1/2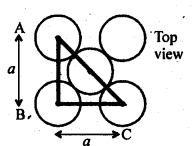 Therefore, 4r = a(2) 1/2 => a = 4r/( 2) 1/2 Thus, Volume of unit cell =a 3 = { 4r/( 2) 1/2 } 3 a 3 = 64r 3 /2(2) 1/2 = 32r 3 / (2) 1/2 No. of unit cell in FCC = 4 Volume of four spheres = 4 × πr 3 (4/3) Thus, packing efficiency = [πr 3 (16/3) ] / [32r 3 / (2) 1/2 ] = 0.74 or 74 %
Therefore, 4r = a(2) 1/2 => a = 4r/( 2) 1/2 Thus, Volume of unit cell =a 3 = { 4r/( 2) 1/2 } 3 a 3 = 64r 3 /2(2) 1/2 = 32r 3 / (2) 1/2 No. of unit cell in FCC = 4 Volume of four spheres = 4 × πr 3 (4/3) Thus, packing efficiency = [πr 3 (16/3) ] / [32r 3 / (2) 1/2 ] = 0.74 or 74 % Let the edge length of the cube be ‘a’ and the radius of each particle be r. So, we can write: a = 2r Now, volume of the cubic unit cell = a 3 = (2r) 3 = 8r 3 We know that the number of particles per unit cell is 1. Therefore, volume of the occupied unit cell
Let the edge length of the cube be ‘a’ and the radius of each particle be r. So, we can write: a = 2r Now, volume of the cubic unit cell = a 3 = (2r) 3 = 8r 3 We know that the number of particles per unit cell is 1. Therefore, volume of the occupied unit cell Hence, packing efficiency
Hence, packing efficiency
 (ii) Body-centred cubic
(ii) Body-centred cubic It can be observed from the above figure that the atom at the centre is in contact with the other two atoms diagonally arranged. From ΔFED, we have:
It can be observed from the above figure that the atom at the centre is in contact with the other two atoms diagonally arranged. From ΔFED, we have: Again, from ΔAFD, we have:
Again, from ΔAFD, we have: Let the radius of the atom be r. Length of the body diagonal, c = 4π
Let the radius of the atom be r. Length of the body diagonal, c = 4π or,
or,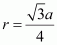 Volume of the cube,
Volume of the cube, A body-centred cubic lattice contains 2 atoms. So, volume of the occupied cubic lattice
A body-centred cubic lattice contains 2 atoms. So, volume of the occupied cubic lattice


 (iii) Face-centred cubic Let the edge length of the unit cell be ‘a’ and the length of the face diagonal AC be b.
(iii) Face-centred cubic Let the edge length of the unit cell be ‘a’ and the length of the face diagonal AC be b. From ΔABC, we have:
From ΔABC, we have: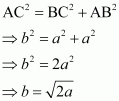 Let r be the radius of the atom. Now, from the figure, it can be observed that:
Let r be the radius of the atom. Now, from the figure, it can be observed that: Now, volume of the cube,
Now, volume of the cube, We know that the number of atoms per unit cell is 4. So, volume of the occupied unit cell
We know that the number of atoms per unit cell is 4. So, volume of the occupied unit cell
 = 74%
= 74% = 107.13 gmol −1 Therefore, atomic mass of silver = 107.13 u
= 107.13 gmol −1 Therefore, atomic mass of silver = 107.13 u It is also given that the atoms of P are present at the body-centre. Therefore, number of atoms of P in one unit cell = 1 This means that the ratio of the number of P atoms to the number of Q atoms, P:Q = 1:1 Hence, the formula of the compound is PQ. The coordination number of both P and Q is 8.
It is also given that the atoms of P are present at the body-centre. Therefore, number of atoms of P in one unit cell = 1 This means that the ratio of the number of P atoms to the number of Q atoms, P:Q = 1:1 Hence, the formula of the compound is PQ. The coordination number of both P and Q is 8.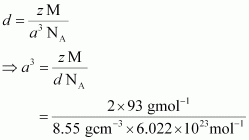 = 3.612 × 10 −23 cm 3 So, a = 3.306 × 10 −8 cm For body-centred cubic unit cell:
= 3.612 × 10 −23 cm 3 So, a = 3.306 × 10 −8 cm For body-centred cubic unit cell: = 1.432 × 10 −8 cm = 14.32 × 10 −9 cm = 14.32 nm
= 1.432 × 10 −8 cm = 14.32 × 10 −9 cm = 14.32 nm A sphere with centre O, is fitted into the octahedral void as shown in the above figure. It can be observed from the figure that ΔPOQ is right-angled ∠POQ = 90 0 Now, applying Pythagoras theorem, we can write:
A sphere with centre O, is fitted into the octahedral void as shown in the above figure. It can be observed from the figure that ΔPOQ is right-angled ∠POQ = 90 0 Now, applying Pythagoras theorem, we can write:
 = 8.97 g cm −3 The measured value of density is given as 8.92 g cm −3 . Hence, the calculated density 8.97 g cm −3 is in agreement with its measured value.
= 8.97 g cm −3 The measured value of density is given as 8.92 g cm −3 . Hence, the calculated density 8.97 g cm −3 is in agreement with its measured value.NCERT Solutions for Class 12 Chemistry Chapter 1 Benefits
There are several benefits to studying the Chapter 1 Class 12 Chemistry NCERT solutions if you want to get good grades.
-
Conceptual Clarity: These solutions don't just teach you facts; they also explain how crystal structures and flaws work, which will help you answer questions that require you to apply what you've learned.
-
Exam Alignment: Most board examinations and competitive tests, like JEE and NEET, use the NCERT curriculum directly to produce the question papers.
-
Time Management: You may quickly check your answers using step-by-step Chemistry Class 12 Chapter 1 solutions without having to look through a lot of books.
-
Visualisation Aid: The way cubic systems are divided into pieces makes it easier to envision how atoms are placed in 3D, which is often the hardest part of this chapter.
-
Accuracy: These NCERT solutions for Class 12 Chemistry Chapter 1 make sure you use the right units (such changing pm to cm) and the right value of Avogadro's constant, which lowers the likelihood of making mistakes when you do math.
Check Out: Class 12th Question Banks
NCERT Solutions for Class 12 Chemistry Chapter 1 FAQs
What are F-centres in the NCERT solutions for Class 12 Chemistry Chapter 1?
F-centres are anionic sites occupied by unpaired electrons. They impart colour to the crystals; for example, they make NaCl appear yellow.
How does temperature affect the conductivity of semiconductors in Chapter 1 Chemistry Class 12 NCERT solutions?
As temperature increases, more electrons can jump to the conduction band, which increases the conductivity of semiconductors.
What is the coordination number in an FCC structure?
In a face-centred cubic (FCC) or cubic close-packed (CCP) structure, the coordination number is 12.
Which defect changes the density of a crystal in Chapter 1 The Solid State?
The Schottky defect decreases the density because ions are missing from the lattice, whereas the Frenkel defect does not change the density.
Why is glass called a supercooled liquid?
Glass is an amorphous solid that has a tendency to flow very slowly over time, similar to liquids, which is why it is termed a supercooled liquid.











