Amines NEET Notes 2026 Exam: Key Topics and Important Questions for Exam Preparation

For aspirants looking for amines NEET notes, this chapter is a goldmine because it connects perfectly with other topics like polymers NEET notes and chemistry in everyday life NEET notes. Understanding how a simple ammonia molecule swaps its hydrogen atoms for alkyl or aryl groups is the key to mastering this section.
The NEET 2026 exam will likely test your conceptual clarity on the basicity of amines and their distinctive chemical tests. While many students get lost in massive textbooks, these amines NEET short notes focus on the reactions and trends that actually show up on the OMR sheet. Let’s break down the nitrogen chemistry you need to know.
Check Out: NEET Books
What is the Structure of Amines?
Amines are derivatives of ammonia (NH_3). Depending on how many hydrogen atoms are replaced by organic groups, they are classified into three types:
-
Primary (1^\circ): One hydrogen replaced (e.g., CH_3NH_2).
-
Secondary (2^\circ): Two hydrogens replaced (e.g., (CH_3)_2NH).
-
Tertiary (3^\circ): All three hydrogens are replaced (e.g., CH_3)_3N.
The nitrogen atom in amines is sp^3 hybridised, leading to a pyramidal shape. Due to the presence of a lone pair, the bond angle is slightly less than the ideal tetrahedral angle of 109.5°.
Physical Properties of Amines (NEET Quick Revision)
-
Lower aliphatic amines are usually colourless and have a fishy smell.
-
With increasing carbon atoms, the physical state commonly shifts from gases to liquids to solids.
-
Aryl amines (like aniline) may appear colourless initially but become yellowish/brown on standing due to slow air oxidation.
-
This kind of observation is useful in NEET theory-based and statement-based questions.
To strengthen your overall NEET preparation and gain more exam-level practice, you can also solve questions from the 39 Years NEET PYQ Physics book, which helps improve problem-solving skills and familiarity with NEET question patterns across previous years.
Physical Properties of Amines Table for Revision
|
Property |
Quick NEET Note |
Why It Matters |
|
Colour/Appearance |
Lower amines are generally colourless |
Helps in basic identification. |
|
Odour |
Fishy or pungent ammonia-like smell |
Common observation-based MCQ point. |
|
Physical State Trend |
Lower: Gases; C3-C11: Liquids; C12+: Solids |
Easy elimination clue in MCQs for state changes. |
|
Aryl Amines Storage |
Often become coloured due to atmospheric oxidation |
A practical fact frequently tested in "Reasoning" questions. |
|
Boiling Point Trend |
1° > 2° > 3° Amines (due to degree of H-bonding) |
Crucial for ranking questions; links with reactivity trends. |
|
Solubility |
Lower aliphatic amines are soluble in water |
Solubility decreases as the molar mass/hydrophobic part increases. |
Boiling Point + H-Bonding
-
Primary and secondary amines can form stronger intermolecular hydrogen bonding because they have N-H bonds.
-
Tertiary amines do not have an N-H bond, so they show weaker self-association.
-
This is why tertiary amines often show lower boiling points than comparable 1°/2° amines.
Quaternary Ammonium Ions
-
Beyond tertiary amines, nitrogen can bond to four alkyl/aryl groups to form a quaternary ammonium ion.
-
In this form, nitrogen carries a positive charge.
-
This is a useful classification extension for NEET revision and makes your notes look more complete.
Check Out: NEET Test series
Practice Important Questions of The Amines Important Questions for NEET
Here are some practice questions to help students test their understanding of amines. The remaining questions, along with detailed explanations and solutions, are available in the downloadable PDF.
Q1
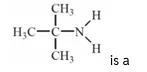
is a
- 1° amine
- 2° amine
- 3° amine
- 4° amine
Q2 A secondary amine is;
- a compound with two —NH₂ group
- a compound with two C-atoms and a -NH₂ group.
- a compound with —NH₂ group on the carbon atom is number two position.
- a compound in which two of the hydrogens of NH₃ have been replaced by alkyl or aryl groups.
Q3 In given compound, all are aryl amines, except;
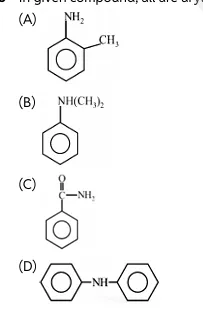
Q4 C₃H₉N represents
- primary amine
- secondary amine
- tertiary amine
- all of these
Q5
CH₃ - C - NH₂
||
O
KOH → (A)
CH₃ - C - NH₂
||
O
LAH → (B)
Relation between (A) & (B) is
- Chain isomer
- Homologous
- Functional isomer
- Identical
Q6
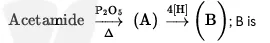
- CH₃NH₂
- CH₃CH₂NH₂
- CH₃NC
- CH₃COOH
Q7CH₃CH₂CONH₂ NaOH Br₂ → A
- Turns blue litmus to red
- Turns red litmus to blue
- Does not affect the litmus
- Decolourise the litmus
Q8 Which of the following statement is correct?
- Methyl amine is slightly acidic.
- Methyl amine is less basic than ammonia.
- Methyl amine is less basic than dimethyl amine.
- Methyl amine is less basic than aniline.
Check Out: NEET Revision Book
Important Topic In Amines NEET Notes
To excel in the 2026 exam, your notes or Amines NEET notes PDF should highlight these specific name reactions:
-
Hoffmann Bromamide Degradation: This is a favourite among examiners. An amide reacts with Br_2 and KOH to give a primary amine with one less carbon atom.
-
Gabriel Phthalimide Synthesis: Used strictly for preparing primary aliphatic amines. Note that aromatic primary amines (like Aniline) cannot be made this way because aryl halides do not undergo nucleophilic substitution with the phthalimide anion.
-
Reduction of Nitriles and Amides: Using LiAlH_4 or catalytic hydrogenation to convert -CN or -CONH_2 into -CH_2NH_2.
4. The basicity of amines is also one of the most frequent topic in Amines NEET notes. Amines are basic because of the lone pair on the nitrogen atom.
-
Gas Phase: Basicity follows the simple inductive effect: 3^\circ > 2^\circ > 1^\circ > NH_3.
-
Aqueous Phase: Here, the trend changes due to solvation effects and steric hindrance.
-
For Methyl substituted amines: 2^\circ > 1^\circ > 3^\circ > NH_3.
-
For Ethyl substituted amines: 2^\circ > 3^\circ > 1^\circ > NH_3.
Check Out: NEET Question Banks
Chemical Tests for Distinction in Amines NEET
Distinguishing between 1^\circ, 2^\circ, and 3^\circ amines is a core part of class 12 amines NEET short notes.
1. Carbylamine Reaction (Isocyanide Test)
Only 1^\circ amines (both aliphatic and aromatic) give this test. When warmed with chloroform (CHCl_3) and alcoholic KOH, they produce a foul-smelling substance called isocyanide or carbylamine.
2. Hinsberg’s Test
Using Benzenesulphonyl chloride (C_6H_5SO_2Cl):
-
1^\circ Amine: Forms a precipitate soluble in alkali.
-
2^\circ Amine: Forms a precipitate insoluble in alkali.
-
3^\circ Amine: Does not react.
Formula of Diazonium Salts
Aryl diazonium salts (ArN_2^+X^-) are vital intermediates. They are prepared by the reaction of Aniline with NaNO_2 + HCl at 0–5°C. They are used in:
-
Sandmeyer Reaction: To introduce -Cl, -Br, or -CN into the benzene ring.
-
Gattermann Reaction: Using Cu powder instead of Copper salts.
-
Coupling Reactions: To form brightly coloured Azo-dyes (important for chemistry in everyday life NEET notes).
Since reactions involving diazonium salts are frequently asked in NEET, students should revise their mechanisms and applications regularly. Along with Chemistry preparation, maintaining strong Physics concepts is equally important. Resources such as NEET Class 11th Physics Med Easy book can help students strengthen their overall NEET preparation through concept-based learning and revision.
Difference Between Various Amine Reactivity
Refer to the table below to understand the basic difference between various amine reactivities.
|
Property |
Primary (1∘) |
Secondary (2∘) |
Tertiary (3∘) |
|
Hinsberg Reagent |
Soluble in KOH |
Insoluble in KOH |
No Reaction |
|
Carbylamine Test |
Foul Smell |
No Reaction |
No Reaction |
|
Nitrous Acid (HNO_2) |
Gives N_2 gas |
Forms Yellow Oily Nitrosoamine |
Forms Nitrite Salts |
|
Boiling Point |
Highest (Max H-bonding) |
Intermediate |
Lowest (No H-bonding) |
Read More: NEET 2025 Toughest Chapters in Physics, Biology and Chemistry
Amines NEET Notes 2026 Exam FAQs:
1. Why is aniline less basic than ammonia?
In aniline, the lone pair on the nitrogen atom is delocalised into the benzene ring due to resonance. This makes the lone pair less available for donation to a proton compared to ammonia.
2. Can we prepare Aniline using Gabriel Phthalimide Synthesis?
No. This method requires a nucleophilic attack by the phthalimide ion on an alkyl halide. Aryl halides do not undergo this reaction easily because of the partial double bond character in the $C-X$ bond.
3. What is the "Isocyanide Test"?
The Isocyanide test (Carbylamine reaction) is used to detect $1^\circ$ amines. When reacted with CHCl_3 and KOH, a primary amine produces an extremely foul-smelling isocyanide.
4. How do amines relate to "Chemistry in Everyday Life"?
Many drugs and alkaloids are amines. For example, adrenaline and ephedrine contain amino groups and are used to increase blood pressure. Benadryl, a common antihistamine, also contains an amino group.
5. Which has a higher boiling point: 1^\circ or 3^\circ amines?
1^\circ amines have higher boiling points because they have two hydrogen atoms available for extensive intermolecular hydrogen bonding. 3^\circ amines have no hydrogen atoms attached to nitrogen, so they cannot form H-bonds with each other.











